(2024-03-15) Unlocking the potential of TiO2-based photocatalysts for green hydrogen energy through water-splitting: Recent advances, future perspectives and techno feasibility assessment
| Author: Obaid F. Aldosari; Ijaz Hussain; |
| Journal: International Journal of Hydrogen Energy (Publication Date: 2024-03-15) |
| Journal Tags: |
| Local Link: Aldosari and Hussain - 2024 - Unlocking the potential of TiO2-based photocatalysts for green hydrogen energy through water-splitti.pdf |
| DOI: 10.1016/j.ijhydene.2024.01.306 |
| Abstract: Hydrogen is becoming more widely accepted as a potential energy carrier due to zero emissions, superior energy capacity, and ecological sustainability. It can be produced in a number of ways, but photocatalytic water splitting using sunlight has recently attracted attention as a sustainable option. Photocatalysts based on semiconductors, especially TiO2 photocatalysts, have been the subject of extensive study because of their desirable physicochemical properties. There are still obstacles to overcome, however, including a wide bandgap, sluggish electron-hole recombination, and the potential for excessive H2 generation. Numerous strategies such as doping, defect engineering, dye sensitization, and semiconductor coupling have been investigated with the goal of improving the performance of TiO2 by discovering solutions to these limitations. This article summarizes current research on the multiple parameters affecting the photocatalytic process in dynamic H2 generation. Surface area, particle size, TiO2 loading, pH, temperature, light source, light intensity, sacrificial reagents, and band gap energy are all important characteristics of photocatalysts. The techno-feasible analysis, current challenges, possibility for subsequent research, and the potential for H2 production by the photocatalytic water-splitting process are further addressed in this article. |
| Note Date: 2024-09-10, 6:44:50 p.m. |
📜 Research Core
Tips: What was done, what problem was solved, innovations and shortcomings?
⚙️ Content
💡 Innovations
🧩 Shortcomings
🔁 Research Content
💧 Data
👩🏻💻 Method
🔬 Experiment
📜 Conclusion
🤔 Personal Summary
Tips: What aspects did you question, how do you think it can be improved?
🙋♀️ Key Records
📌 To be resolved
💭 Thought Inspiration
Nhu dau buoi
Go to annotation “Another way of utilizing visible light to produce hydrogen analogous to dye sensitization is semiconductor composition, also known as semiconductor coupling.” (Aldosari and Hussain, 2024, p. 9)
Go to annotation “Another way of utilizing visible light to produce hydrogen analogous to dye sensitization is semiconductor composition, also known as semiconductor coupling.” (Aldosari and Hussain, 2024, p. 9) Nhu dau buoi
Go to annotation
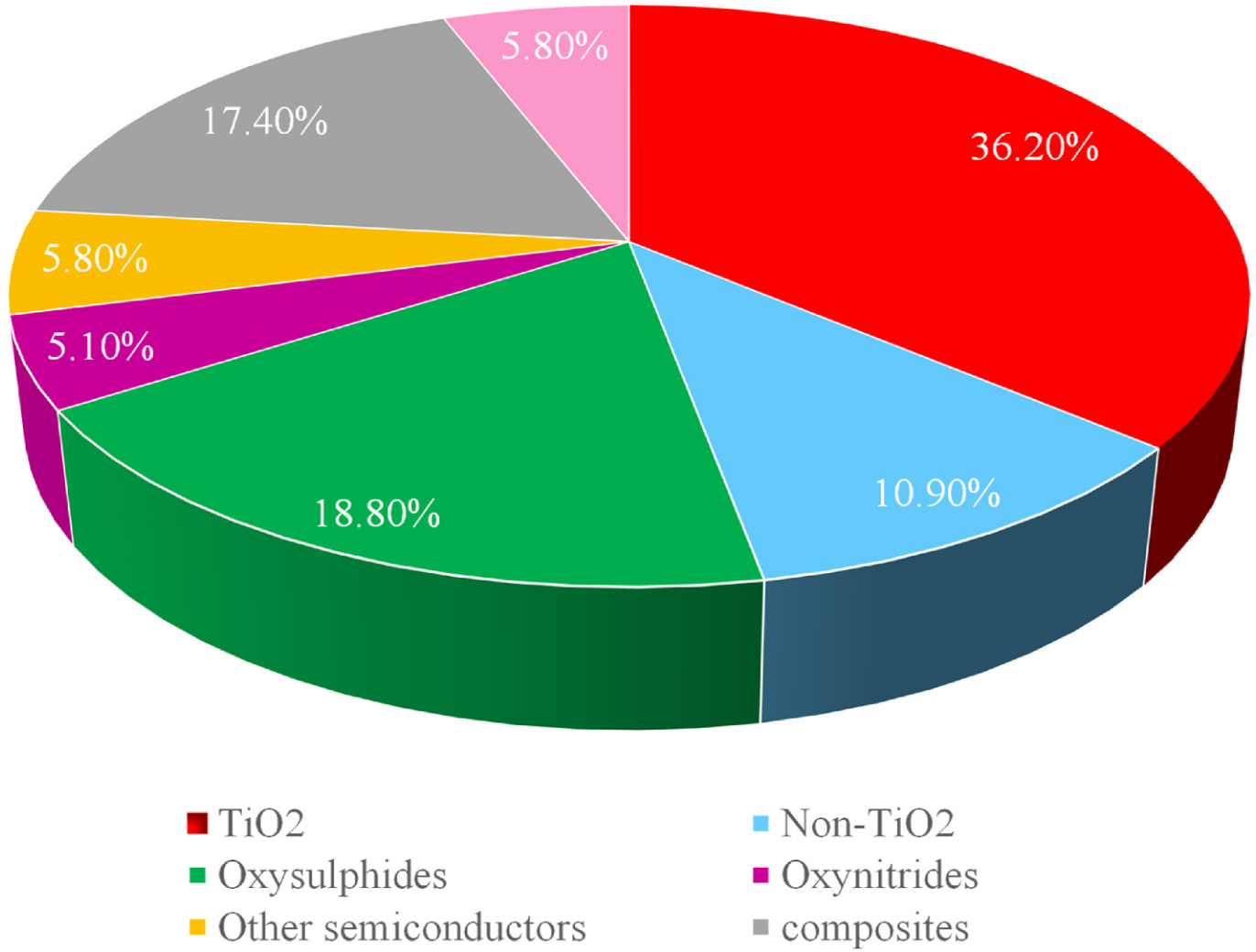
(Aldosari and Hussain, 2024, p. 2) Fig. 1. A statistical analysis of the numerous articles published on photocatalytic H2 production through water splitting over many different ph
Go to annotation
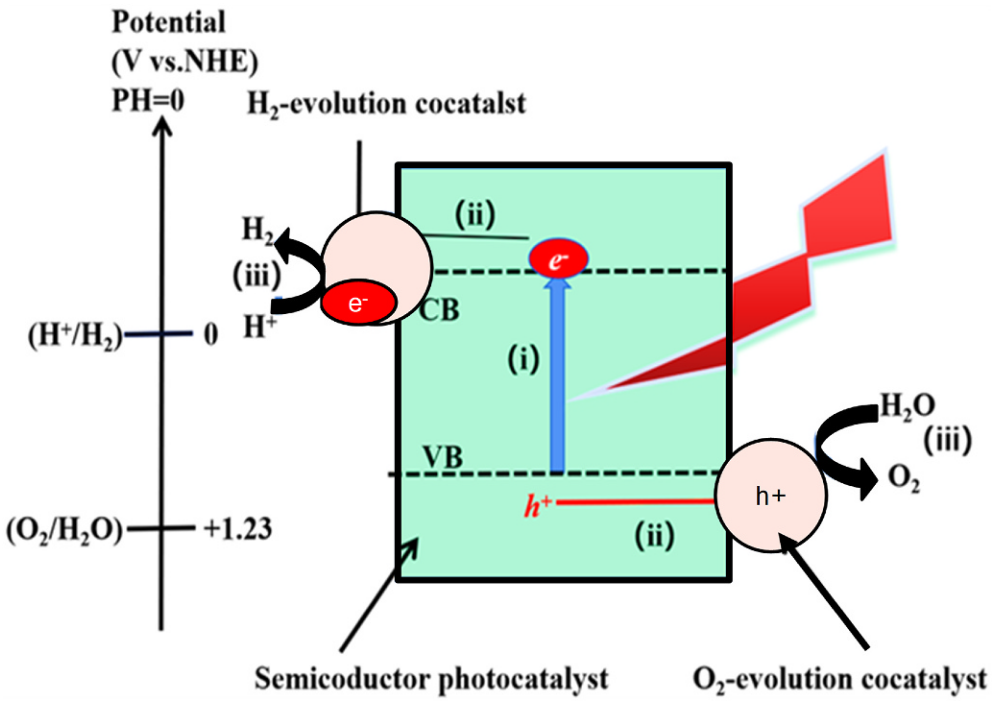
(Aldosari and Hussain, 2024, p. 3) Fig. 2. A schematic diagram shows the underlying idea behind photocatalytic water splitting.
otocatalytic materials. Adapted from Ref. [23].
Go to annotation
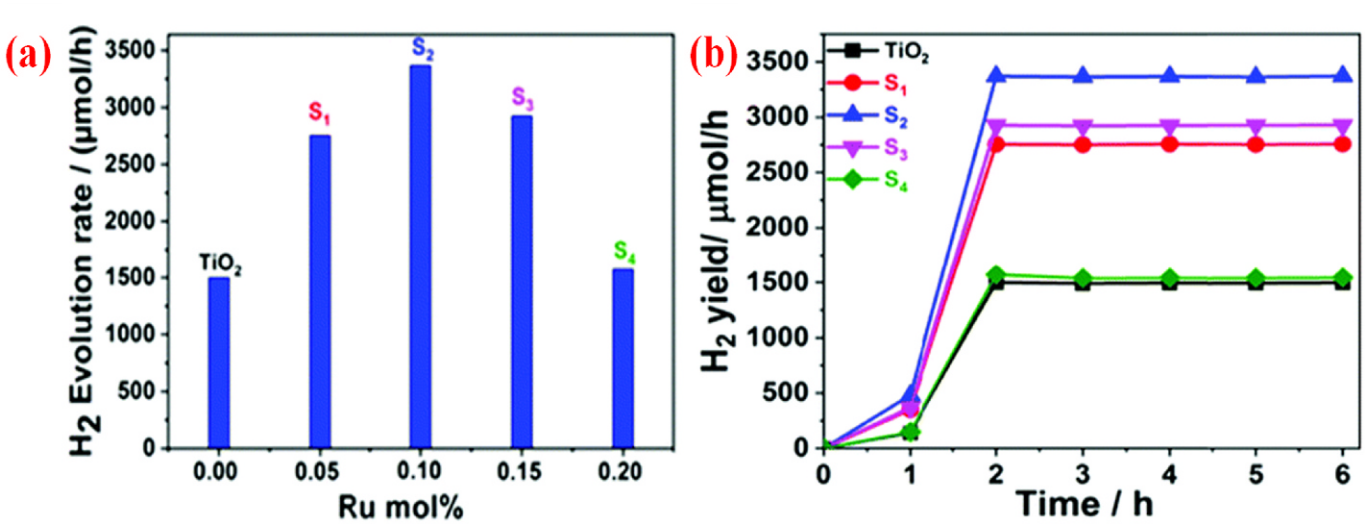
(Aldosari and Hussain, 2024, p. 6) Fig. 5. (a) The rate at which hydrogen produced under light of ? ? 320 nm for un-doped TiO2 and other TiO2 samples containing Ru. (b) Photocatalytic H2 production over un-doped TiO2 and different Ru doped TiO2 samples under light of ? ? 320 nm for 6 h. Reprinted with permission from Ref. [58].
Go to annotation “Dye sensitization is essential for harvesting visible solar radiations for efficient energy conversion and boosting hydrogen evolution alongside metal and nonmetal ion doping and composite production [110]. The redox property and visible light sensitivity of certain dyes make them useful in solar cells and photocatalytic systems [111]. Excited electrons in the dye jump from the HOMO level to the LUMO level, where they can then be transported to the valence band of TiO2 [112]. Absorbing visible light efficiently and having excited dyes transfer electrons to the CB of TiO2 efficiently lead to a high hydrogen generation rate, as shown in Fig. 10 [113]. It has been discovered that hydrogen can be produced with just visible light active dyes and no semiconductors at all [114]. Dyes like safranin, O/EDTA, T/EDTA, and others have been studied for their potential to detect visible light. There have been reports of a few dyes that are capable of producing hydrogen in the absence of semiconductors; however, in the absence of an efficient charge separation mechanism, the majority of the excited electrons recombine with the holes, resulting in a decrease in the synthesis of hydrogen’s overall efficiency [115].” (Aldosari and Hussain, 2024, p. 9)
Thanh yeu My
- My mung
- Yjp
- Thanh rat yeu My